The Rise of Genetic Testing, and the Missing Piece
Over the last decade, genetic testing has exploded.
Clinicians now have access to more patient data than ever before:
- Direct-to-consumer DNA kits
- Clinical genomic panels
- Advanced biomarker integrations
On the surface, this should have transformed care.
But in reality, something hasn’t kept up.
Access to data has increased. Clinical utility has not.
Most providers already feel this gap in practice.
A patient walks in with a report—or you order one yourself—and after reviewing pages of markers, SNPs, and risk scores, the real question emerges:
“Now what?”
This is the core failure point of modern genetic testing.
The Problem: Data Without Direction
Most genetic testing platforms deliver information—not decisions.
They provide:
- Long SNP lists
- Static PDF reports
- Generalized associations
But they stop short of what clinicians actually need: clear clinical direction.
This creates a significant burden inside real-world practice.
The Reality for Clinicians
- Non-actionable reports → Data without treatment pathways
- Conflicting interpretations → Different tools suggest different meanings
- Trial-and-error protocols → Guesswork replaces precision
- Time burden → Hours spent interpreting instead of treating
The result is predictable
Clinicians either:
- Spend excessive time trying to interpret data
- Or stop using genetic testing altogether
And the promise of precision medicine stalls.
As outlined in the EndoDNA clinical framework, the issue isn’t access to genetics—it’s the lack of structured interpretation and application
Why SNP-Level Thinking Falls Short
At the core of the problem is how genetic data is interpreted.
Most platforms still rely on SNP-level analysis:
- Single gene variants
- Isolated risk markers
- One-to-one associations
But biology doesn’t work this way.
Genes Don’t Operate in Isolation
Human physiology is driven by interconnected systems, not individual markers.
SNP-level thinking fails because it:
- Ignores pathway interactions
- Misses cross-system effects
- Oversimplifies complex biology
For example:
- Hormone metabolism is influenced by multiple genes interacting together
- Neurotransmitter balance depends on entire pathways—not single variants
- Detoxification, inflammation, and metabolism are deeply interconnected
Focusing on isolated SNPs is like trying to understand a city by looking at a single street.
You miss the system entirely.
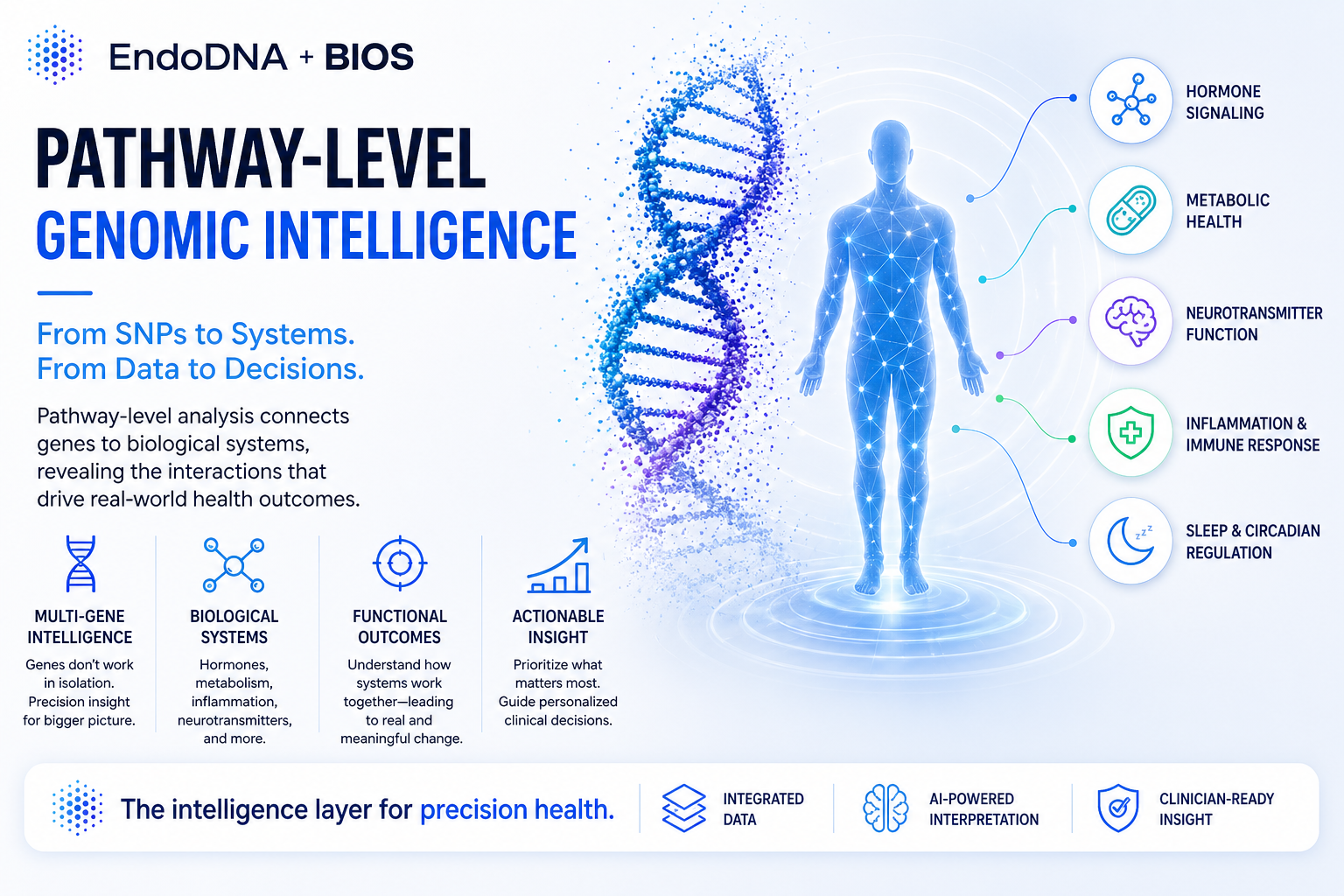
The Shift to Pathway-Level Intelligence
To make genetic testing clinically useful, interpretation must evolve.
This is where pathway-level intelligence changes everything.
Instead of analyzing genes individually, this approach:
- Maps genetic variants to biological pathways
- Connects interactions across systems
- Models how the body actually functions
What This Looks Like in Practice
Pathway-level analysis organizes genetics into systems such as:
- Hormone metabolism and signaling
- Inflammation and immune response
- Neurotransmitter function
- Metabolic and insulin pathways
- Detoxification processes
This creates something fundamentally different:
A structured understanding of how a patient’s biology behaves—not just what variants they have.
EndoDNA is built specifically on this model—translating genomics into pathway-based biological insight rather than isolated markers
From Report → Decision: What Clinicians Actually Need
For genetic testing to be valuable in practice, it must support decision-making—not just analysis.
Clinicians need:
1. Clarity Around Symptoms
Why is this patient experiencing what they’re experiencing?
2. Structured Treatment Direction
What should I actually do next?
3. Confidence in Personalization
Can I explain and defend this approach to my patient?
Without these three elements, genetic testing becomes:
- Interesting
- Informative
- But ultimately unusable
The gap isn’t data.
It’s translation.
How EndoDNA + BIOS Solves the “Now What?” Problem
EndoDNA + BIOS was built specifically to close this gap.
Instead of delivering static reports, the platform functions as a clinical intelligence layer—turning complex biological data into structured, actionable insight.
What Makes It Different
- Pathway-Level Interpretation
Organizes genetic data into biological systems, not isolated SNPs - AI-Powered Clinical Decision Support
Translates complexity into prioritized treatment pathways - Structured Outputs
Clear, clinician-ready summaries—not overwhelming reports - Real-Time Clinical Guidance
Supports decisions during actual patient consults - Integrated Data View
Combines genetics, labs, and patient history into one system
The result:
Clinicians move from interpretation → action.
Instead of asking “What does this mean?”
They can ask:
“What do I do next?”
And get a clear answer.
As defined in the platform architecture, BIOS serves as the translation layer between genomics and clinical care—organizing complex data into usable clinical insight
The Future of Genetic Testing Is Clinical Intelligence
Genetic testing is not going away.
But the way it’s used is changing.
The next evolution isn’t:
- More reports
- More data
- More markers
It’s better decision support.
The Future Looks Like:
- Systems that think in biological pathways
- Platforms that learn from patient outcomes
- Tools that reduce—not increase—clinical workload
- Intelligence layers that support real-time decisions
In other words:
Not genetic testing platforms.
Clinical intelligence platforms.
That’s the shift.
And it’s already happening.